Dangerous Substances and Dust Explosions: An Introduction
With any risk assessment, the first step is to identify the hazard. When working with dangerous substances such as flammable liquids and gases, hazard communication is usually straightforward. Containers carry standardized hazard pictograms, and the Safety Data Sheet (SDS) provides detailed information on the properties, handling requirements, and ignition characteristics of the material. For example, gasoline or acetone will be labeled as flammable, and the SDS will specify their ignition conditions and safe use guidelines.
Below are examples of toxic and flammable hazard pictograms.

Flammable Hazard Pictogram

Toxic Hazard Pictogram
When working with dangerous substances such as gasoline or acetone, both the container and the Safety Data Sheet (SDS) provide clear hazard information. Labels identify the material as flammable, and the SDS outlines its physical and chemical properties, safe handling practices, and ignition conditions. This data helps facilities assess fire and explosion risks when storing or using volatile liquids.
Combustible Dust and the SDS
For combustible powders, hazard communication is often incomplete. Unlike liquids and gases, powders capable of causing a dust explosion do not carry standardized pictograms. Packaging for common materials such as flour, sugar, or powdered additives does not indicate that these substances can combust.
This lack of labeling is problematic for developing process safety strategies in both industrial and everyday environments. While a Safety Data Sheet (SDS) is provided in industry, the information is frequently limited. In many cases, the SDS will only note that a material “may combust” without supplying the ignition or explosibility data required to design protective measures.
Without quantitative testing of particle size, ignition sensitivity, and explosion severity, SDS information alone is insufficient for assessing the true hazard. This gap often leaves facilities without the technical basis needed to evaluate and control combustible dust risks.
What is Combustible Dust?
Combustible dust is any finely divided solid that can become suspended in air, ignite, and propagate an explosion when exposed to an ignition source. In this state, even materials not normally considered flammable can create a significant hazard.
Materials commonly classified as combustible dust include:
Powders, flakes, fines, and fibers
Organic solids such as flour, grain, sugar, and wood
Carbonaceous materials such as coal, charcoal, and soot
Textile fibers such as cotton
Metals including aluminum, magnesium, and iron filings
Certain inorganic powders
The explosibility of dust depends largely on particle size and distribution. Powders finer than approximately 500 microns disperse readily in air, forming explosive atmospheres that are highly sensitive to ignition sources such as sparks, hot surfaces, or electrostatic discharge.
This behavior explains why materials that appear non-combustible in bulk form can burn or explode once reduced to a fine dust. The combination of dispersion, oxygen, and ignition conditions provides the environment for a dust explosion to occur.
U.S. Standards for Combustible Dust
In the United States, combustible dust hazards are regulated under OSHA and NFPA standards.
NFPA 652 requires facilities to complete a Dust Hazard Analysis (DHA) to identify where combustible dust hazards exist and what safeguards are needed.
NFPA 499 provides guidance on Hazardous Area Classification (HAC) for gases, vapors, and dusts, determining where ignition controls are required.
NFPA 654 covers prevention of fire and dust explosions in facilities handling combustible particulates.
Avoiding a Flammable or Explosive Atmosphere
A flammable or explosive atmosphere forms when fuel and oxygen are present in the right proportions and exposed to an ignition source. This concept is illustrated by the fire triangle, which consists of:
Fuel
Oxygen
Ignition source
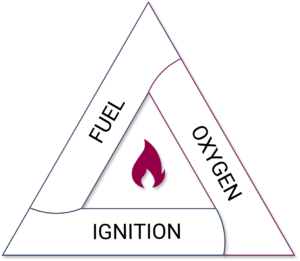
Fire Triangle
For example, methane gas confined in a pipe is not flammable because oxygen is absent, even though the container is labeled “extremely flammable.” If that methane leaks into the atmosphere, it mixes with air to form a combustible mixture that can ignite and explode.
The same principle applies to dust explosions. When fine particles disperse in air, they form a flammable atmosphere highly sensitive to sparks, embers, or hot surfaces. Controlling dispersion and confinement is therefore central to preventing dust explosions.
Avoiding Ignition in Explosive Atmospheres
If a combustible atmosphere cannot be prevented, ignition sources must be controlled. In the U.S., this is addressed through Hazardous Area Classification (HAC) studies, following NFPA 499. These assessments define hazardous zones and determine where specialized electrical and mechanical equipment must be installed to reduce ignition risk.
Preventing Injury from Ignition Events
Even with prevention and ignition control, ignition may still occur under certain conditions. In these cases, protective measures such as explosion venting, suppression, or containment systems are applied to reduce the consequences of a dust explosion. These safeguards are part of the wider framework required by NFPA 652 and referenced in OSHA enforcement.
The Explosion Pentagon
The explosion pentagon expands on the fire triangle by adding two additional elements:
Dispersion – a dust cloud formed by mixing fuel particles with air
Confinement – an enclosure that allows pressure to build
When dispersion and confinement are present along with fuel, oxygen, and an ignition source, the result is a dust explosion capable of generating destructive overpressure.
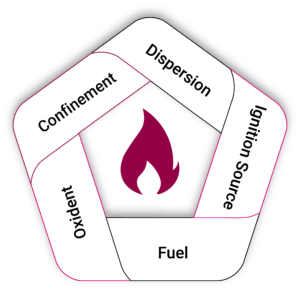
If fuel and air are mixed in the open, a flash fire may occur, but pressure does not build. In contrast, when combustion happens inside confined spaces such as silos, ducts, or processing vessels, the flame front accelerates and transitions into an explosion.
Materials, Chemical Reactions and Combustion
Combustion is an oxidation reaction in which a material reacts with oxygen. If a material is not fully oxidized, it is capable of further reaction with oxygen. This reaction is exothermic, releasing heat that can produce visible flames.
In most cases, liquids and solids do not combust directly. Instead, vapors and gases generated from heating undergo oxidation when mixed with air. The reaction between these vapors and oxygen produces heat, flame propagation, and pressure rise.
For dusts, the principle is similar. Finely divided particles present a large surface area for oxidation. When dispersed in air, these particles behave like vapors and gases, allowing rapid combustion. Under confinement, this behavior leads to a dust explosion rather than simple burning.
Solid Materials and Surface Area
Many substances used in industries such as food processing and pharmaceuticals are not classified as flammable in bulk form but can combust when reduced to fine powders.
For solids, combustion occurs at the surface where oxygen can react with the material. The surface area-to-volume ratio determines how readily this reaction takes place. A larger surface area allows faster oxidation, increasing the rate of combustion.
This relationship explains why dust explosions are strongly dependent on particle size distribution. Powders with smaller particle sizes present more surface area and are more reactive in air. As a general reference, materials finer than about 500 microns are typically considered combustible dusts capable of producing explosive atmospheres.
Dust Explosion Tests: Dust Combustibility Test
To determine whether a powder is classified as a combustible dust, laboratories perform a dust combustibility test, sometimes called an explosibility screening.
In this method, a small sample of the material is placed in a chamber where a controlled pulse of air disperses it into a dust cloud. Electrodes then introduce a strong electrostatic spark to act as the ignition source.
If ignition occurs, the material is classified as combustible and capable of producing a dust explosion under the right conditions.
If ignition does not occur, the material is considered non-combustible in dust cloud form.
This screening provides a yes/no answer and serves as the first step in characterizing dust explosion hazards before more advanced testing, such as determining Pmax or Kst.
Test Preparation
Before performing a dust combustibility test, solid materials must be prepared according to standardized methods. Samples are typically sieved to below 500 microns, since particle size is a primary factor in determining explosibility.
If a bulk powder contains mostly large particles with only a small fraction of fines, an “as-is” sample may not ignite because there is insufficient combustible dust to sustain combustion. For this reason, sieving ensures that the fine fraction responsible for dust explosions is properly evaluated. Larger particles are less likely to produce a positive combustibility result.
Suppliers and Material Data
Supplier documentation often states that a material is “not combustible.” However, when tested under proper laboratory conditions, the same material may exhibit combustible behavior. This discrepancy usually arises because testing was performed on an unsieved bulk sample. Without isolating the fine fraction, the test does not reflect the true explosibility of the dust.
Accurate classification therefore depends on testing conducted according to recognized standards, which specifically require sieving and controlled dispersion. Only then can the results provide a reliable basis for assessing dust explosion hazards.
Material Examples: Iron Bar
A pure iron bar is not combustible. It can be heated, shaped, or melted in a furnace, but it will not ignite or sustain a flame. However, when the same iron bar is ground into iron filings, its behavior changes. Exposed to a flame, filings will combust because their particle size allows rapid oxidation.
If reduced even further into a fine powder, iron can become pyrophoric — igniting spontaneously upon exposure to air. This example illustrates how particle size distribution directly affects combustibility and the potential for a dust explosion. An ignition source is still required, but the finer the material, the lower the energy needed to ignite it.
Combustible Dust and Industrial Processes
In many operations, bulk materials may not appear hazardous because they do not form airborne dust clouds under normal conditions. However, attrition during processing can reduce particle size and generate combustible dust.
Examples include:
Milling, grinding, or polishing
Conveying and transfer points
Dust extraction and collection systems
In processes where fine particles are created or deliberately separated, explosive atmospheres can form. When these atmospheres are exposed to ignition sources, the conditions for a dust explosion are met.
Conclusion: Why Test Dust Particles?
Testing powders and particulate materials is essential for determining whether they qualify as combustible dusts capable of producing a dust explosion.
If testing shows that a material is not combustible, additional protective measures may not be required.
If the material is combustible, the data becomes the foundation for Dust Hazard Analyses (DHA) under NFPA 652, Hazardous Area Classification (HAC) studies under NFPA 499, and the engineering of explosion protection systems.
For U.S. facilities, combustible dust hazards represent both a safety and regulatory obligation. NFPA 652 requires revalidation of DHAs every five years, and OSHA may issue citations under the General Duty Clause when combustible dust risks are not adequately addressed.
Accurate laboratory testing, combined with DHA and HAC studies, provides the technical basis for safe process design, regulatory compliance, and long-term operational reliability.



